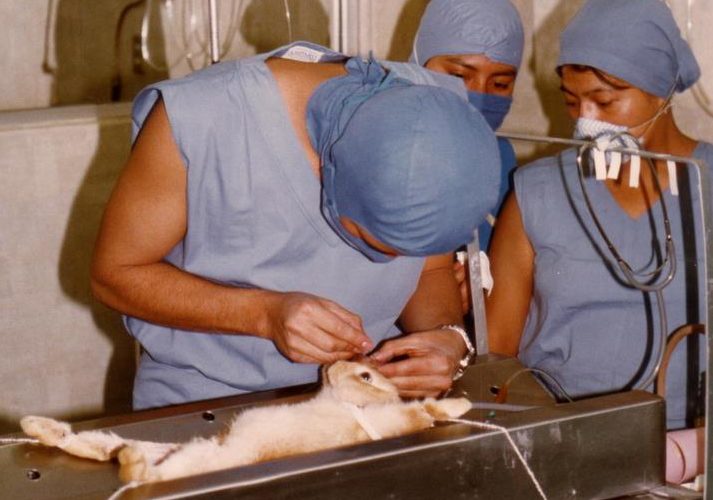
The controversial “Draize” irritation tests that measure eye and skin irritation using rabbits are banned in several countries.
In a major victory for animal rights campaigners, the Union Health Ministry has prohibited use of the cruel and obsolete “Draize” irritation tests that involve using rabbits.
The test was developed more than 70 years ago to measure eye and skin irritation using rabbits, who are locked in restraints while a test chemical is applied to one eye or to the shaved skin on their backs. Animals are monitored for up to two weeks, without pain relief, for signs of chemical damage, which can include swelling, ulceration, bleeding and blindness.
This move comes after Union Minister for Women & Child Development, Maneka Gandhi, wrote to the Ministry regarding the practice, followed by representations Humane Society International (HIS) to end the obsolete animal testing technique, as it did not provide reliable or even relevant information for safe human use.
“This is a historic decision by the health ministry. Thousands of rabbits will now be spared from one of the most cruel and infamous experiments on rabbits known to mankind,” said Alokparna Sengupta, deputy director of HSI/India.
“This, in addition to the cosmetic testing ban passed in India in 2014, will provide the much-needed impetus for India to move towards more humane and advanced 21st century science. We laud the Health Ministry for acting swiftly to notify the rule change and hope that the companies follow this in letter and spirit.”
The ministry notification, dated November 4, 2016, mandates the use of Organization for Economic Cooperation and Development (OECD) validated non-animal alternatives. The decision comes after a series of representations made by HSI/India, People for Animals and other stakeholders, informing the Central Drug Standard Control Organization and the Drug Controller General (India) regarding internationally recognized alternatives.
A variety of validated and internationally recognized non-animal alternatives, including reconstructed human skin and corneal tissues, have been available for years – some since 2004. These 21st century technologies, deployed as part of a sequential testing strategy, allow for complete replacement of the Draize eye and skin tests with methods that are more reproducible and reflective of human responses.
The Hindu